|
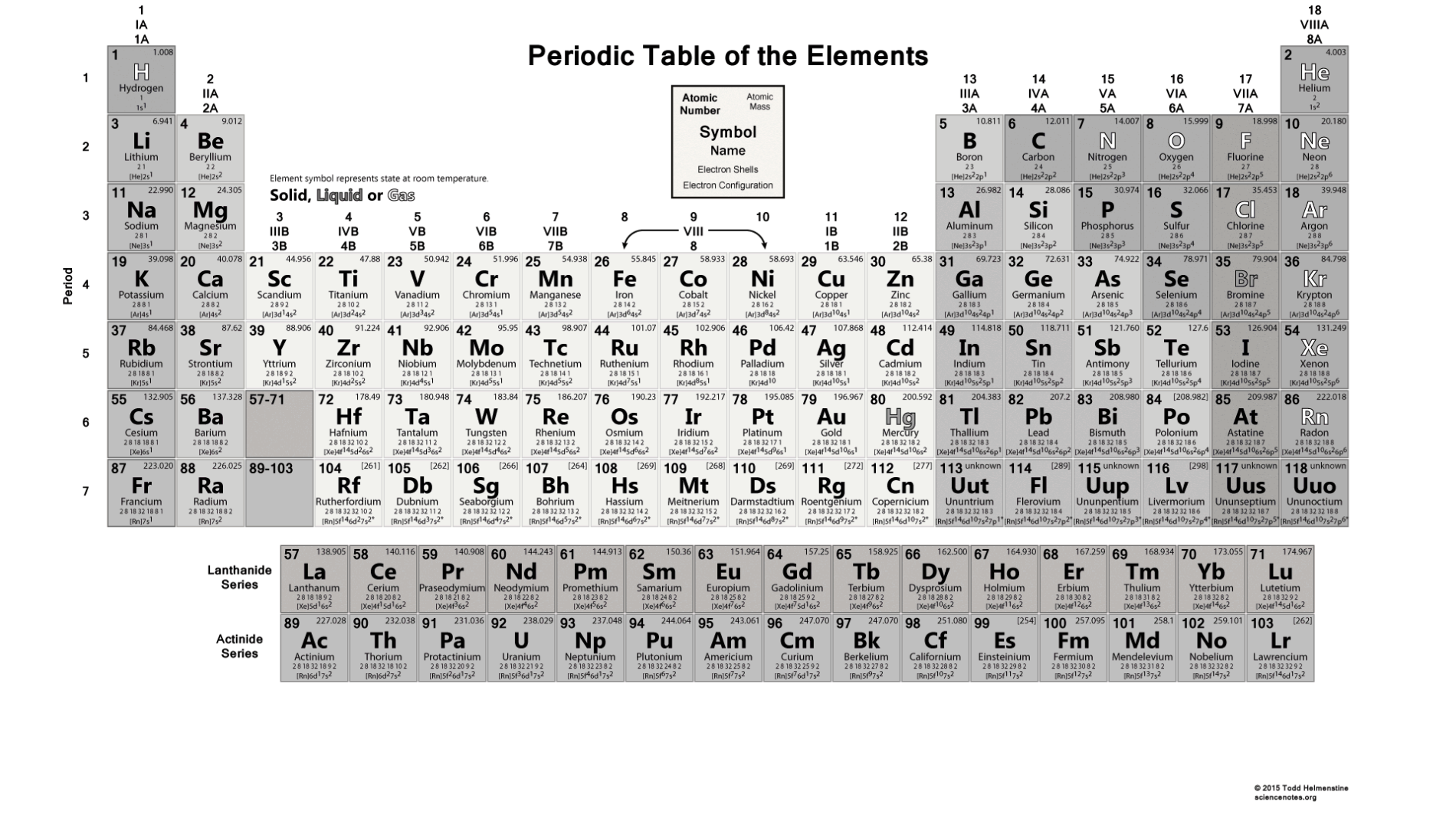
Now, remember, isotopes are atoms with the same number of protons but different numbers of neutrons and therefore, the difference in mass numbers comes from the different number of protons. If you are given data stating that the atomic mass of potassium is, for example, 39, then this is referred to that specific isotope. This is the average atomic mass of all the isotopesof potassium and that is why it is not a whole number. Next, on the button, we have the average atomic mass of potassium which is equal to 39.098. 
There cannot be another element with the atomic number 19. So, potassium has 19 protons, and this is true for all its isotopes. This is the number usually given on the top of the symbol, and, in this case, it is 19. The first, you want to identify is the atomic number of the element because this is unique for each element. For example, if we look up potassium, K in the periodic table, we will see the following numbers: Atomic weights of elements with atomic numbers 110-116 taken from this source.Before we start with some examples of calculating the number of protons, neutrons, and electrons, let’s recall what information we get about a particular element from the periodic table. IUPAC Standard Atomic Weights Revised (2005).Atomic weights of elements with atomic numbers from 1-109 taken from this source. Atomic Weights of the Elements 2001, Pure Appl.Note 5: The atomic weight of commercial Lithium can vary between 6.939 and 6.996-analysis of the specific material is necessary to find a more accurate value.Ĭhemical series of the periodic table references:.Note 4: The isotopic composition varies in terrestrial material such that a more precise atomic weight can not be given.Note 3: The isotopic composition of the element can vary in commercial materials, which can cause the atomic weight to deviate significantly from the given value.Note 2: The isotopic composition of this element varies in some geological specimens, and the variation may exceed the uncertainty stated in the table.However, three elements, Thorium, Protactinium, and Uranium, have a characteristic terrestrial isotopic composition, and thus their atomic mass given. , indicates the mass number of the longest-lived isotope of the element. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
